EndoDraft® – Surgical Planning Software
for Endovascular Interventions
🛠️ EndoDraft is a lightweight, standalone surgical planning software designed to assist interventional radiologists and vascular surgeons in planning physician-modified endografts (PMEGs). It enables precise design of fenestration locations, visual feedback in 2D/3D, and exports print-ready plans for intraoperative use.
Now available: Advanced 3D Spatial Planning Module (Beta)
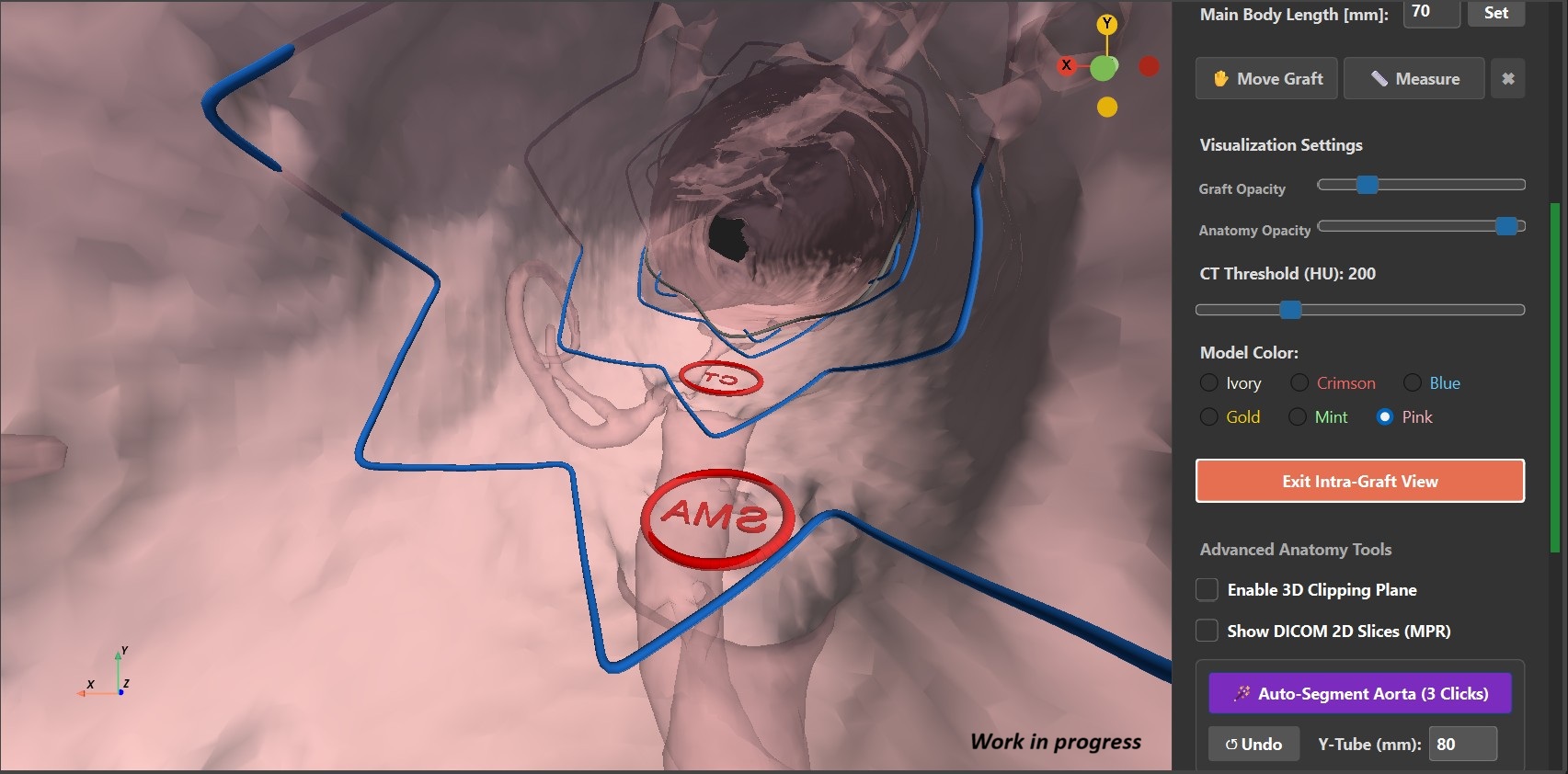
The new 3D spatial planning module is available! Load patient CT scans directly, reconstruct accurate isosurface models, perform automated aortic segmentation, position the graft model, perform custom edits and verify fenestrations via an internal lumen view.
📰 Published in Journal of Endovascular Therapy
A Novel Surgical Software Tool to Improve the Physician-Modified Endograft Workflow
Bendegúz Juhos, Csaba Csobay-Novák et al. — We present EndoDraft, a dedicated software solution designed to streamline the planning and creation of fenestrated endografts.
🎥 Demo


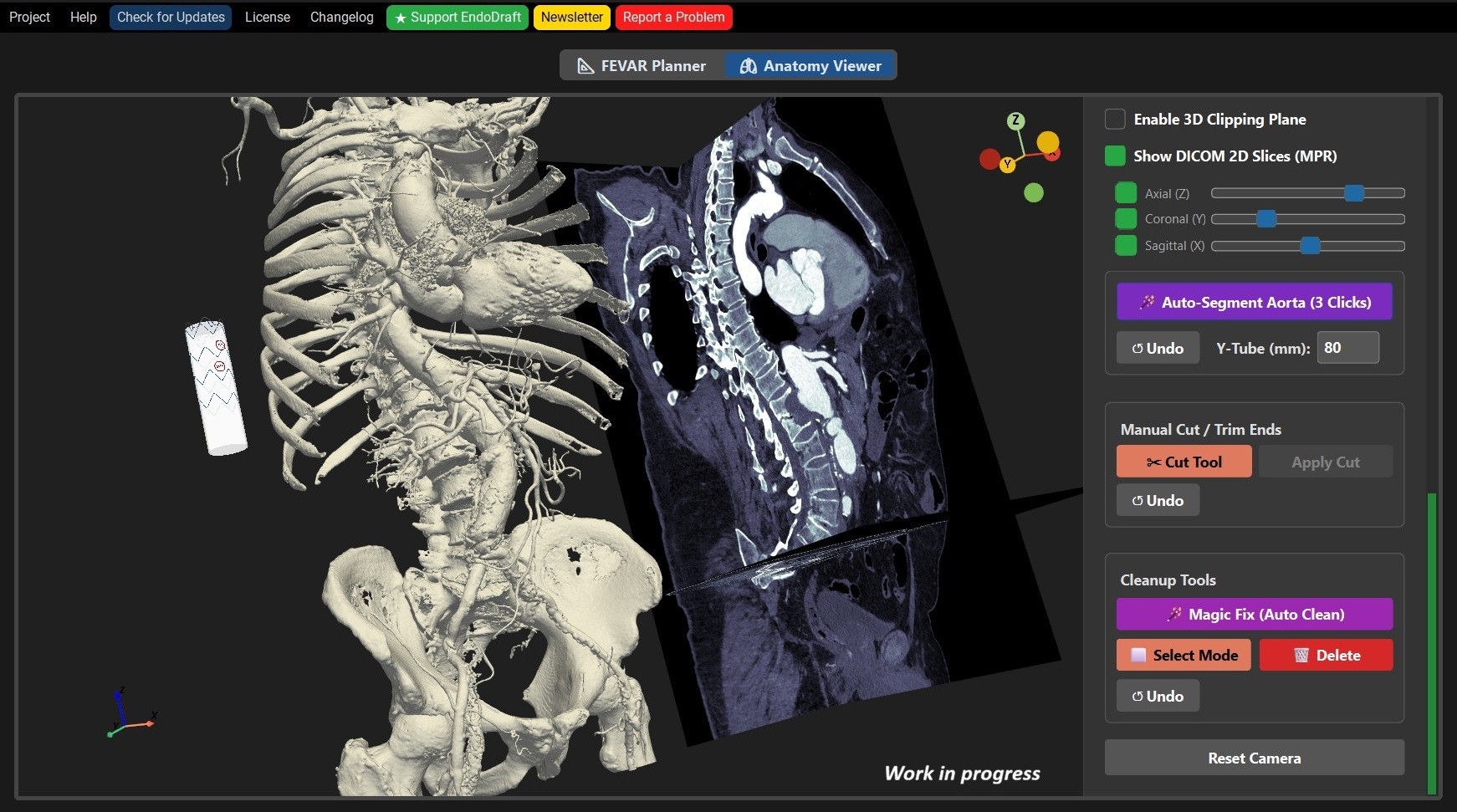
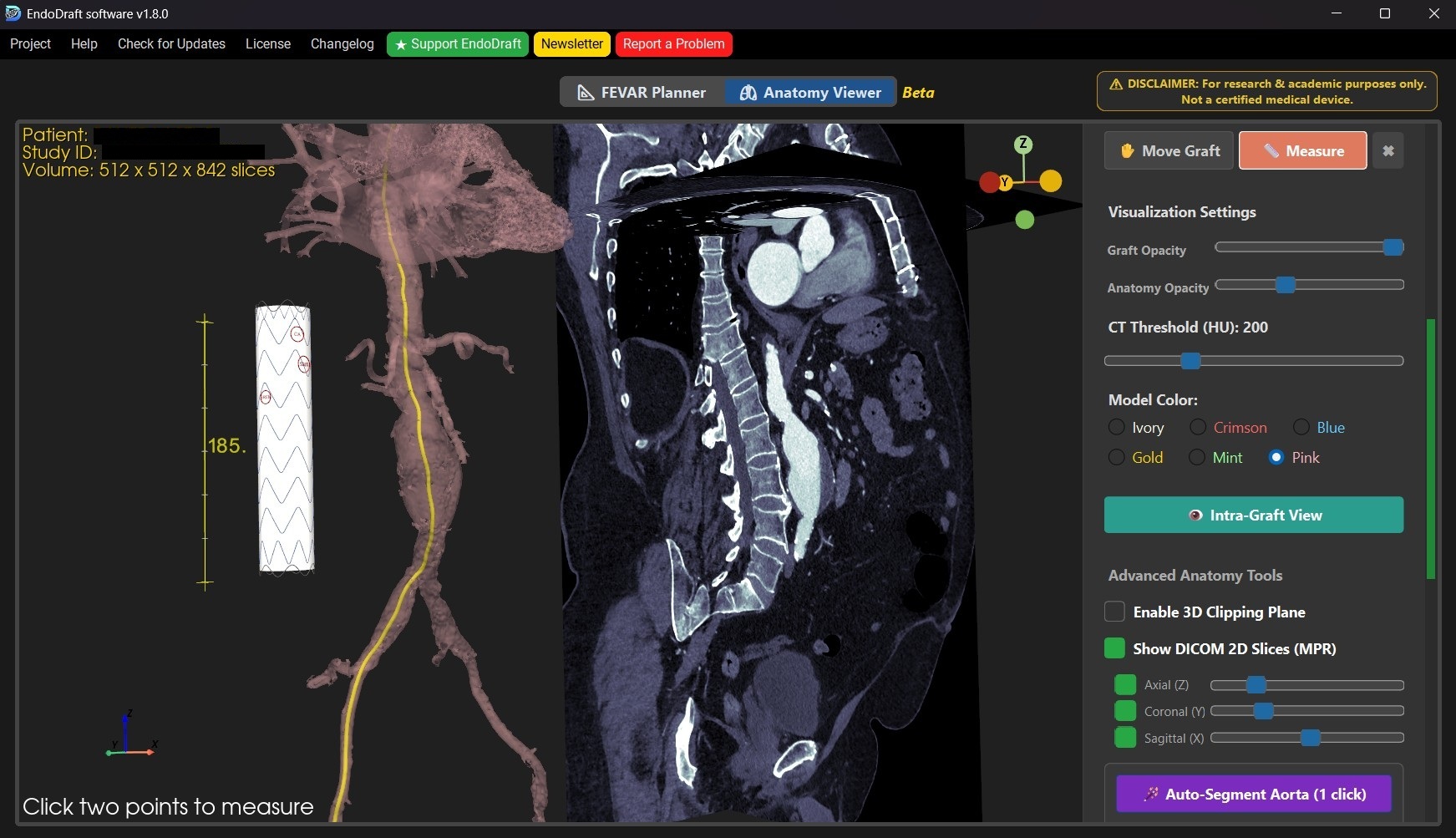
Screenshots while using the software.
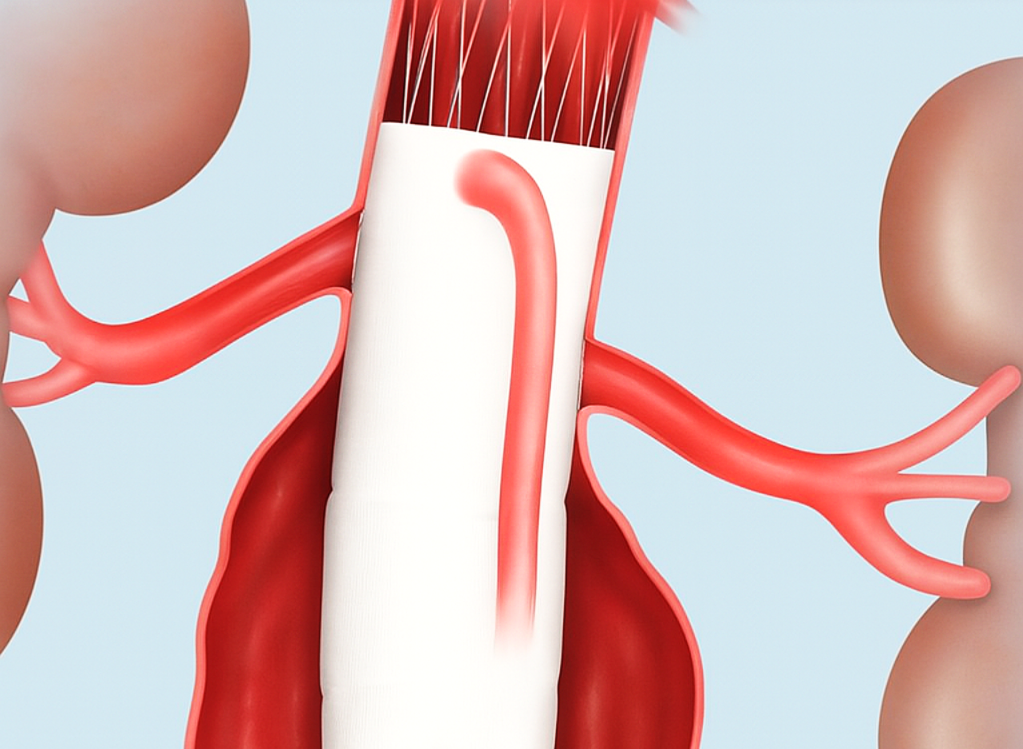
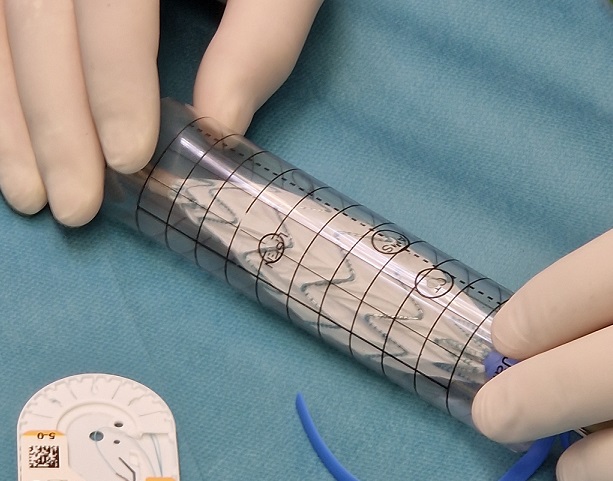
Sample graphics and photo about deploying a laser printed punch card made in EndoDraft.
🚀 Powerful Features
A complete toolkit for the precise preoperative planning of Endovascular Interventions: EVAR, FEVAR, PMEG.
Native DICOM Engine
Load raw CT scans directly. Visualize anatomy with real-time Multi-Planar Reconstruction (MPR), Hounsfield thresholding, and dynamic clipping planes.
1-Click Segmentation
Advanced voxel-based morphology isolates the aorta, removes spine/bone artifacts, and generates highly accurate anatomical centerlines in seconds.
3D FEVAR Planning
Map target vessels directly on the 3D anatomy. EndoDraft automatically calculates tortuosity-aware arc lengths and precise clock-face projections.
Smart Graft Fitting
Snap and bend straight tubular endograft models seamlessly along the patient's anatomical centerline to verify fenestration alignment.
Interactive 2D Templates
Drag-and-drop fenestrations on an unrolled graft map with real-time delta measurements and commercial strut collision visualization.
Surgical Export
Generate print-ready PDF reports with precise axis scaling. Supports automated G-Code generation for sterile laser cutting.
📦 Download
By downloading and using the software, you agree to the Terms of Use.
⚠️ Windows SmartScreen & Antivirus Notice
Since the software does not yet have a digital signature (it costs a lot!), some operating systems (especially Windows) may display a warning when launching the .exe file, such as: “Windows protected your PC” or “Unknown publisher”.
This is standard behavior for unsigned applications and does not mean the file is dangerous. EndoDraft has been thoroughly tested and is totally safe to use, so you can ignore the warning.
✅ If this happens, to proceed on Windows:
- Click “More info”
- Then select “Run anyway”
For security reasons, installation is required with EndoDraft_Setup_vX.X.X.exe.
📝 Release Notes
Full history on GitHub: Releases
🖥 System Requirements
📧 Feedback or Contact
If you have questions, feature requests, or feedback, feel free to contact us directly on LinkedIn:

Bendegúz Juhos MSc
Electrical Engineer MSc, PhD Fellow clinical medicine
Department of Interventional Radiology
Semmelweis University

Csaba Csobay-Novák MD, PhD
Interventional Radiologist, PMEG professional
Department of Interventional Radiology
Semmelweis University
OR
📜 License / Terms of Use
© 2026 Bendegúz Juhos
- EndoDraft® is provided free of charge for academic, educational and personal use only.
- Redistribution, modification, or commercial use of the software is strictly prohibited without prior written permission from the author.
- The software is distributed as a compiled executable (
.exe), with installer/setup file. No source code is provided. - The software collects anonymous usage statistics (e.g., graft type, number of fenestrations, OS version) to support development and research.
- NO PATIENT DATA, names, or identifiable personal information are transmitted. All data processing is strictly anonymous.
- The software is provided “as is”, without warranty of any kind, express or implied.
⚠️ Legal Disclaimer / Medical Device Notice
EndoDraft® is an experimental research tool provided exclusively for use by trained vascular specialists to support their own planning workflow. It is not a certified medical device (no CE mark, FDA approval, or equivalent) and does not provide medical advice, diagnosis, or treatment. It must not be relied upon as a standalone tool for direct clinical decision-making.
The clinician remains solely responsible for all clinical decisions, medical procedures, and the use of any modified endografts.
Use is entirely at your own risk and subject to the full Terms of Use / Legal Notice.
By using this software, you agree to the terms above.